The topic of fuels for fusion is much talked about in the fusion start-up community. What fuel are you using is a common question, particularly from US VC investors. Yet it is barely discussed in the publicly funded fusion programmes, neither magnetic nor inertial. This post is the first in a series where I will outline why it is not discussed in the mainstream of fusion. To me, the answer is simple; it is because deuterium-tritium is the only viable fuel. I will present the analysis and you can make up your own mind.
What are the options?
Let’s start at the beginning. The sun is our solar systems resident fusion reactor, why are we talking about options for fuel? Isn’t the objective to build a mini-sun, don’t we just follow the example? Not exactly. The sun fuses hydrogen, normal hydrogen with one proton as the nucleus and nothing else. And eventually the hydrogen turns into helium, natures second element, but the path to get there is quite complicated. The overall process is called the proton-proton chain.
The rate-limiting step in this process is the first one, the fusion of two protons together to form a deuterium nucleus, an isotope of hydrogen with a proton and a neutron, and it is a spectacularly difficult fusion reaction. Protons in the sun wait around for 9 billion years, on average, before eventually fusing. Because of this very slow reaction, the heat output of the sun per unit volume is actually very low, less than the metabolic heat output of a person and about the same as a compost heap. The sun makes up for its poor fusion performance by being truly enormous.
To make fusion work on Earth we need a fuel with much higher energy density, it can’t be hydrogen. There are four candidates commonly discussed. The first is deuterium and tritium (DT), which fuse to produce a helium-4 nucleus, also called an alpha particle, and a neutron. Next there is deuterium and helium-3 (DHe3), which also produces an alpha particle but now with a proton and no neutron. Then there is the pure deuterium case (DD), where deuterium fuses with itself. This reaction has two branches, both of which occur with equal probability. The first produces tritium and a proton, and the second helium-3 and a neutron. And lastly there is normal hydrogen again, but in this case fusing with boron-11, producing three alpha particles.
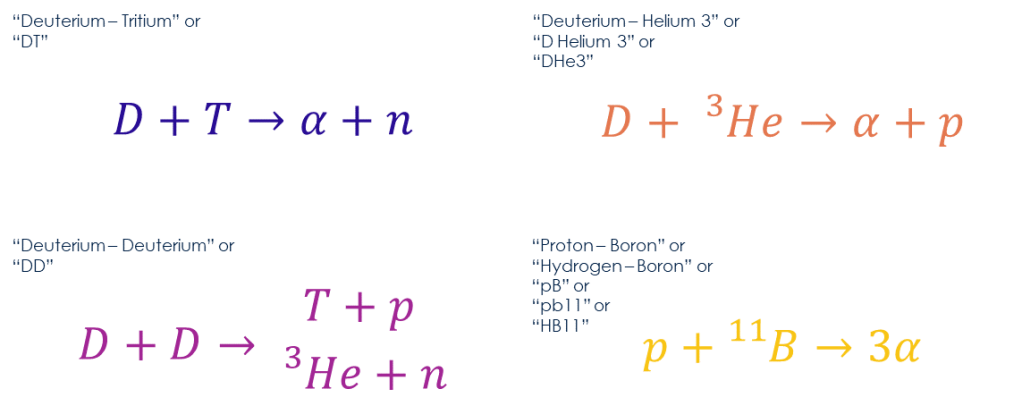
These four are the reactions with the highest fusion cross section. The cross section describes the likelihood of fusion occurring as a function of the centre of mass energy of the two reactants. Imagine an experiment where you fire two atomic nuclei, two ions, at each other at a specified velocity. The cross section is the chance that they fuse, which depends on the velocity of the two ions. In fact, this is exactly how experiments to measure the fusion cross section are done, using a particle accelerator. Don’t get it confused with CERN though, the ion energies needed are far less than those needed for particle physics.
For application to fusion power the cross section gets turned into something called the reactivity. Fusion power needs a plasma in thermal equilibrium, a requirement that I will write about in future but not as part of this series, and in a plasma in thermal equilibrium, the ion energies follow a Maxwellian distribution. The ions are in a dynamic equilibrium, they are constantly flying around and constantly bumping into other ions. Sometimes they fuse but most of the time they “scatter”, they bounce off. If we follow an individual ion as it goes on its journey inside the plasma, it would be constantly undergoing collisions. Sometimes it would be going slowly and get whacked by a fast-moving ion, speeding up as a result. Sometimes it will be the fast ion, losing energy to its slower compatriots. It’s a bit like being stuck inside a big crowd.
Overall, looking at all the ions, there is a distribution of different velocities present. Any individual ion will be bouncing around all over, sometimes fast, sometimes slow. But the net result is a population with a clearly defined average energy and a fixed set of velocities present. If the temperature is higher, the average energy is higher, and the more ions we have at the highest velocities. Actually, formally, this should be said the other way around. This is the definition of temperature. When we say a plasma has a particular temperature, what this actually means is we have ions with a particular Maxwellian distribution of velocities; having this distribution defines the temperature.
And this dynamic equilibrium isn’t just a plasma thing, nor this definition of temperature. The molecules in the air around you are constantly bashing into each other in exactly the same way, and the velocities will follow the Maxwellian distribution for room temperature. The average nitrogen molecule bumping into your skin right now is moving at about 940 mph.
Coming back to fusion, the cross section tells us the probability of fusion for a given velocity, and the temperature tells us the velocities we have in the fuel. To find out how much fusion we get, we integrate and count up the contribution of each specific velocity to the total reaction rate. The quantity that results from this integration is called the reactivity.
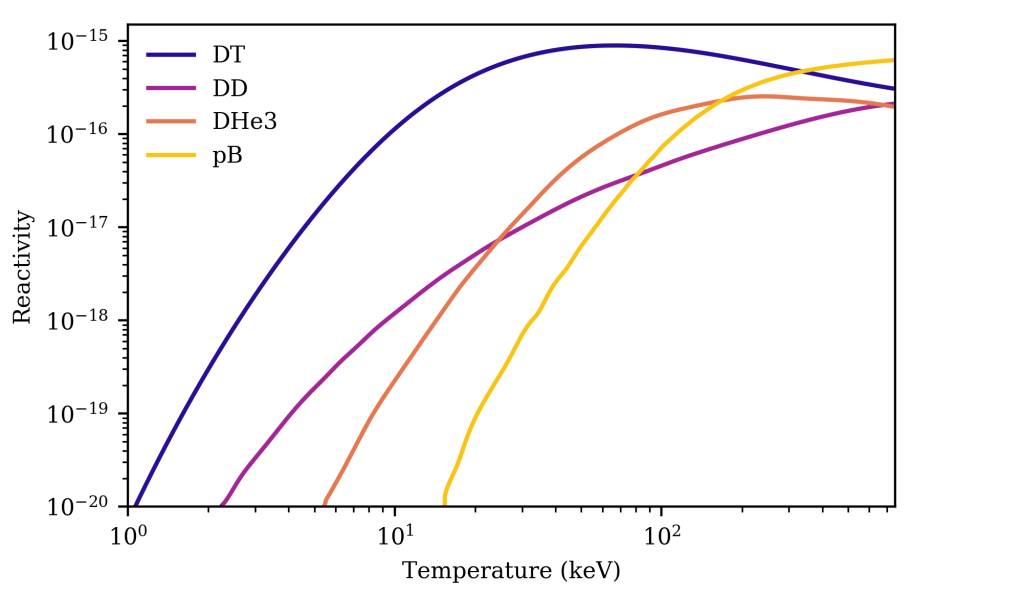
Plotting the reactivities allows the different candidate fuels to be compared. The first thing that we can see is that DT has the highest reactivity at nearly every temperature, and that the DT reactivity ramps up at the lowest temperature. The second-best reactivity is initially DD, until a temperature of about 24 keV, after which DHe3 takes over. This itself is then surpassed by pB at a temperature of 167 keV, which eventually surpasses DT at 335 keV, after DT has gone past its peak. The DT peak is the biggest number overall, however.
(For reference, 1 keV is about 10,000,000 K)

We can also plot these reactivities a bit differently. The first plot has a logarithmic scale on both axis, which can make even major differences look small. Instead, I’ve plotted them as a ratio of the DT reactivity. This shows the difference very clearly. At lower temperatures, the easiest part of parameter space, the part we want to be in, the DT reactivity is more than 50 times higher than the alternatives. It isn’t until a temperature of 64 keV that DHe3 comes within even 10% of the DT value.
The reactivity tells us how many reactions are happening per unit volume per unit time, and this is not exactly what we want. What we really want is energy, and that means we need to look at the energy released by each reaction. For DT, a single reaction liberates 17.6 MeV of energy. For DD, the value is lower, 3.65 MeV. DHe3 releases the largest amount at 18.3 MeV, and then pB gives 8.7 MeV. To account for the differences in energy released, we can scale the reactivities by the appropriate value. Looking at it this way, DT is the best fuel at all temperatures.

Having established that DT is best in terms of power density we now need to discuss the potential benefits of other fuels, and there are potential benefits. First, two of these candidate fuels require only naturally occurring inputs. Both tritium and helium 3 don’t exist naturally on Earth in any useful quantity, they will have to be manufactured to be used as fusion fuel. And second, two of the candidate fuels produce neutrons and two do not. This is important because neutron damage is one of the major engineering challenges of fusion. Both DHe3 and pB produce no neutrons, not directly anyway.
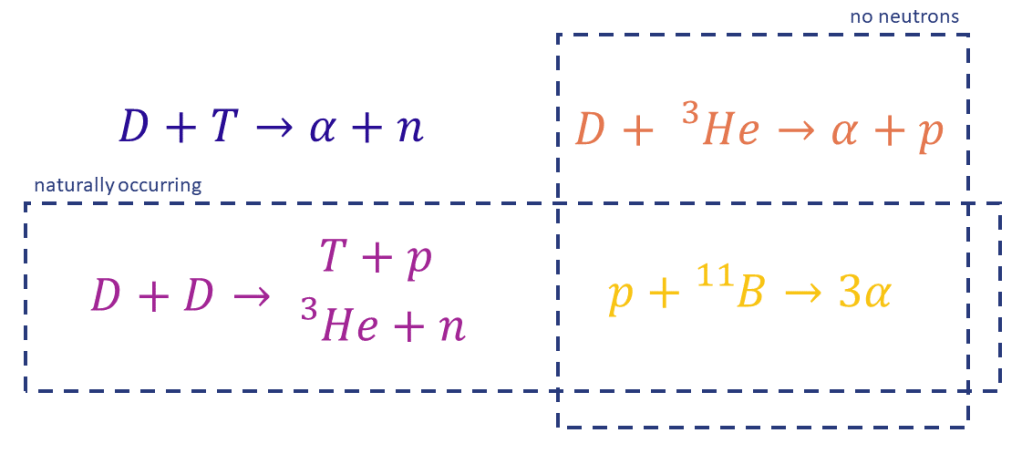
What we have seen here is that DT has the highest power density, but we have also seen that the other fuels do have their advantages. We have not established is what is actually needed for useful power production. It might be that all four options can work great, DT would produce the most power, but the others are fine too. Spoiler alert, this is not the answer, and this is what we will explore in the next few posts.

Thank you for the very clear explanation of this topic which I have seen is also the subject of debate online. As you said there will be further articles so please ignore my questions if you intend to answer them later.
I am particularly interested in Temperature.
1) You talk about fusing temperature in terms of keV but ions created from fusion in terms of MeV which I understand more as kinetic energy. These are the same sorts of units, but off the scale on your fusion rate vs temperature graph. Are these generated ions capable of meaningful fusion at these MeV energies, or must they be slowed down significantly first?
2) Can you tell from your simulations just how high a temperature you think you can repeatably achieve by your projectile method alone?
3) Is it possible to use a little DT or DD fusion to generate the heat to ignite a higher temperature pB11 reaction all in just one of your targets? If so, might that have some advantages in terms of material availablity, cost, equipment lifetime etc. ?
LikeLike
Hi Chris. Thanks for the questions. Let me go point-wise.
1) yes, it is a bit weird expressing kinetic energy of the fusion products in MeV, and then using the same unit for temperature. There is a deep connection, however. When in thermal equilibrium and having a Maxwellian distribution, there is a clearly defined average energy for the ions, which is 3/2 * k * T, where T is the temperature and k is the Boltzmann constant. When working in units of eV for both energy and temperature, k = 1. This means that at, for example, 10 keV temperature, the average kinetic energy is 15 keV. This can be compared directly to the energy released from fusion, which is measured in MeV, i.e. it is far larger. This shows that the process for producing energy can in principle work. I will write more about the energy inputs and outputs in a later post.
http://hyperphysics.phy-astr.gsu.edu/hbase/Kinetic/molke.html
The reactivity has different units. This is number of reactions per unit volume per unit time.
2) in inertial fusion, the heating is achieved by compression, and this indeed can be simulated. We are working with DT and need single figure keV temperatures
3) a very good question, one which I will be writing about. Short answer, DT to light pB, no. DT to light DD, could work
LikeLike
What about the bremsstrahlung losses of the alternative fuels, in particular p-B ?
The book by Parisi and Ball, “The Future of Nuclear Fusion”, has an excellent explanation of this. It seem like these losses make it very unlikely the other fuels could be used.
Do you agree with this?
LikeLike
OK, thanks for this link, I had not seen this Blog entry.
The bottom line seems to imply there is something else coming on p-B. Could you give a clue? Is there any possibility p-B could work?
LikeLike
Just went out this morning, about 2 hours after your comment! I will write more about pB. The analysis so far has 50:50 mix of pB. This gives you the most fusion power but more radiation loss than a boron-poor mix. There is an optimum percentage. I’ll go through this and a few other things in future but my view is that it can’t work
LikeLiked by 1 person
Hi Nick, could you tell me why p-Li7 or D-Li6 are almost never considered ? To me, they look easier than pB11.
LikeLike