The production, or breeding, of tritium is one of the major engineering challenges of fusion. The easiest fusion reaction uses deuterium and tritium; it requires the lowest temperature and can return the most energy. The core reaction fuses one deuterium and one tritium, producing one alpha particle, which is to say a helium 4 nucleus, and one neutron. The alpha particle has one fifth of the energy (3.5 MeV) and the neutron takes the remaining four fifths (14.1 MeV).

This introduces a challenge because tritium is not a stable isotope. The half-life is 12 years; medium length on a human timescale, but nothing on geological or astrophysical timescales. This means that there is no natural abundance, it decays too quickly, and therefore any fusion power plant must produce its own tritium in a closed cycle.
There are many reasons that tritium is challenging. It is not a stable isotope, as in, it is radioactive. It is also hydrogen, which makes it “leaky”, able to percolate through containment structures. And it is also chemically identical to hydrogen, as in, it is flammable. This all complicates the plant required to extract, purify, store, and recirculate produced tritium into the machine. For these reasons tritium is difficult, but these things are solvable. There is, however, a deep-seated nuclear physics constraint that makes closing the fuel cycle, producing enough tritium, very challenging for some fusion power plant designs.
Tritium is produced using a reaction between the neutron from the fusion reaction itself and lithium. The output of this reaction is another helium 4 and one tritium. The problem is that we have used exactly one tritium to make exactly one neutron, to make exactly one tritium. To make this work we would need to capture every single neutron. Every single one must react with lithium and produce tritium. And we’d have to usefully extract every single atom of tritium produced. Neither of these things are possible.

Fortunately, this is still a simplification, one commonly used and it is normally where the story stops. In fact, there are two stable isotopes of lithium, lithium 6 and lithium 7, and the reaction with the neutron is different between the two. With lithium 7, we get a neutron back out again. This neutron can then go on to interact with another lithium and produce a second tritium. For one neutron in, we can get more than one tritium out.

But again, this is STILL a simplification! There are many things that can happen when a neutron collides with a lithium nucleus. The TENDL-2019 database of neutron cross sections has 88 entries for lithium. All of these entries describe a different possible outcome from the event. Only two produce tritium, and only five produce further neutrons, of which the reaction with lithium 7 mentioned above is the most likely. The rest are mainly different types of “scattering”, different ways that the neutron just bounces off the lithium nucleus.
Each of these processes has a different likelihood of occurring, and that likelihood or “cross-section”, depends on the energy of the neutron coming in. The lithium 7 reaction that gives both a neutron and a tritium works well at the “birth” energy of the neutron, 14.1 MeV, the amount of energy it has just after fusing. However, the lithium 7 reaction is endothermic, meaning that the energy of the output neutron is less than the input neutron, which means that the probability of another lithium 7 reaction is lower. You can’t have an endless process going round and round producing more and more tritium each time. And anyway, scattering is overall the more likely outcome, which also lowers the neutrons energy and makes the lithium 7 reaction less likely. This reaction falls off a cliff as the neutron energy goes down, and that is the direction that nature is taking it.
Eventually, at low energies, when we have “thermal neutrons”, the tritium producing reaction with lithium 6 becomes the most likely process, so thermal neutrons produce tritium very well, but this reaction consumes the neutron.
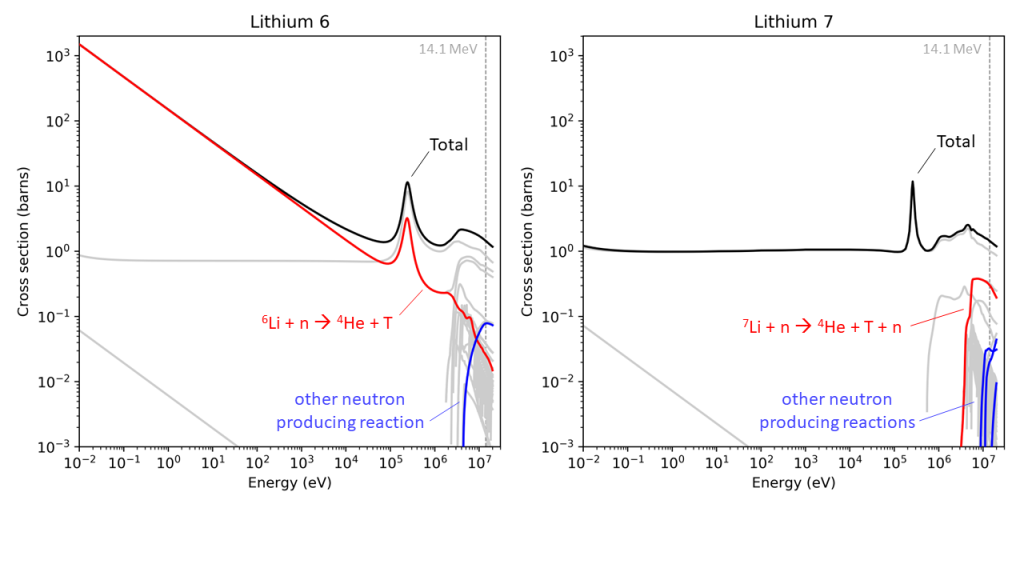
So, thinking of the process as one reaction with lithium 6 and another with lithium 7 is a pretty good approximation. But the outcome of all the detail about energies, cross-sections, and scattering is that there is a limit to the maximum number of tritium atoms one can produce per neutron. In the field this is called the “tritium breeding ratio” and the nuclear physics says it can never be higher than 2. It can be changed by changing the ratio of the two isotopes of lithium, a common proposal for fusion power plant designs struggling to close the loop and produce enough tritium, a very challenging proposal as there is no supply chain for enriched lithium, but it can’t get above this limit of 2.
There are also other ways to introduce neutron multiplication. There are spallation-like reactions with both beryllium and lead that are often discussed. Beryllium is often introduced through the use of FLiBe, a blend of the salts lithium fluoride and beryllium fluoride, acting as a molten salt coolant. And lead is often introduced through the use of lead-lithium eutectic mixture as the coolant. Both of these options lead to the liberation of a neutron from the target nucleus, i.e. the beryllium or the lead, and produce a lighter isotope of the same as the other product. Regardless of all the options, and even combining these with the option of enrichment of the lithium, it is not possible to produce more than two tritium atoms per neutron.
And these additions that can improve the tritium production do not come for free. Beryllium is highly toxic, as is lead. FLiBe introduces fluorine chemistry, and yes, tritiated hydrofluoric acid will be produced. Lead also has a highish atomic number and the range of different isotopes produced rather challenges the “no radioactive waste” statement, polonium being one of the eventual products.
Some suggest using fissionable material, e.g. uranium. The neutron multiplication possible by this route is higher, and the tritium breeding ratio can be greater than two. But why bother? My view is that if you are going to have uranium in your plant, you may as well build a fission plant. You will have all the same difficulties to deal with and fission already works.
This fundamental limit on the amount of tritium that can be produced per neutron is the deep-seated reason that closing the fuel cycle is a challenge. If you cannot capture more than 50% of the neutrons for tritium production, you fundamentally cannot close the loop and be tritium self-sufficient. Space used for other things eats into the total amount of neutrons being captured. In magnetic fusion, neutral beam heaters, RF heaters, the magnets themselves, the central column in a tokamak, the first wall and a host of other things all eat into the space available. In laser inertial fusion, the entrance ports for the many laser beams have the same impact. And in designs that are cylindrical in nature, the ends of the cylinder cannot typically be used for tritium production.
Fortunately for First Light, we can capture 99% of the neutrons in the flowing lithium coolant inside the reaction vessel. We can achieve a tritium breeding ratio of up to ~1.5 simply with normal lithium, no additives, and no enrichment. Any when I say fortunately, what I of course mean is, we deliberately designed it that way.
Learn more here…
https://firstlightfusion.com/technology/power-plant
